Leica TCS SP8 STED Microscope
The purchase of the super-resolution microscope, in the summer of 2017, was partially funded by the National Science Foundation (NSF MRI Program), the New York Capital Region Research Alliance (NYCAP) and Rensselaer Polytechnic Institute (RPI) as part of a joint grant application (RPI, NYS Dept. of Health Wadsworth Center, Albany Medical College, and SUNY Albany). This is the only one of this kind available within 150 mile radius.

The microscope enables mechanistic and morphological studies in biology, biomaterials and tissue engineering due to the instrument’s nanoscale resolution combined with its multiple modalities: live-cell imaging; quantitative studies of expression and/or co-localization; 3D imaging of biomolecules and bio-material morphologies; and dynamic imaging. It will also play a crucial role in the novel application of fluorescent dyes and will be instrumental in visualizing different aspects of tissue engineering.
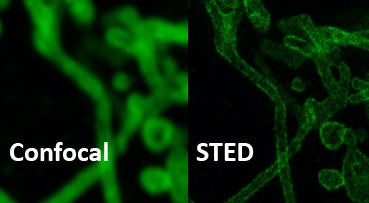
Conventional Confocal and STED (70nm resolution)
The instrument introduces important advances in three crucial areas: super-resolution in all dimensions (STED in 3D), super-resolution over the whole spectrum and in live cell imaging. To accomplish super resolution in the Z dimension the instrument has a second light path with a different phase mask. The multi-color super resolution is achieved with three laser lines: two continuous wave lasers at 592 nm and 660 nm and a pulsed laser at 775 nm. In addition, the White Light Laser, the AOBS (acousto optical beam splitter) and tunable spectral detectors allows any kind of fluorophore combination. To improve resolution with live cell imaging we acquired Gated STED (allows to shift the time gate away from the excitation pulse), that not only increases resolution but less laser power is needed enhancing the viability of the cells. We are also offering light-sheet imaging capabilities.
In addition to the acquisition software provided by the manufacturer, the STED microscope is also equipped with the Huygens Professional software package. This deconvolution program improves resolution, contrast and signal to noise ratio in the images making the analysis more accurate and robust, as well as obtaining statistics of individual objects, even when doing 3D image reconstruction.
The imaging software package Imaris is also available for broader imaging capabilities (in particular when working with three- and four-dimensional multi-channel images of any size).
| RPI Users | External Non-Profit | External Industry | External Industry Partners | |
|---|---|---|---|---|
| Standard | $65 | $107 | $214 | |
| Assisted | $130 | $214 | $428 | |
| Training | $89 | $146 | $293 |
| RPI Users | External Nonprofit | External Industry | External Industry Partners | |
|---|---|---|---|---|
| Personnel Time | $77 | $127 | $253 | $228 |
Effective August 1, 2025 through July 31, 2026. Rates are hourly unless specified otherwise and are subject to change without further notice.
Required Acknowledgement and Authorship
Please acknowledge the CBIS Core Facilities in all publications and grant applications where our equipment and/or personnel have facilitated the work. These acknowledgements are very important because documenting our contributions helps to ensure that the resources of the Core Facilities are sustainable.
- Equipment: If you used Core Facility equipment, please note this in the Materials and Methods. e.g., Thermogravimetric analysis was carried out using a TA Instruments TGA-Q50 (Rensselaer CBIS Analytical Biochemistry Core Facility).
- Personnel: Please consider including CBIS personnel as co-authors on your publications when they have made a significant intellectual contribution to the research. Include CBIS Core Facility directors or staff as co-PI or co-investigators in grant applications when they provide a significant contribution to the grant proposal and scientific/intellectual leadership for the proposed work. Please follow these guidelines: ABRF Recommended Guidelines for Authorship on Manuscripts. Also, our Core Facility personnel always appreciate when they are mentioned in the Acknowledgements section of publications.
- Required Funding Authorization Form: Rensselaer researchers must fill out the CBIS Cores Authorization Form (PDF) to use the CBIS Core Facilities.
